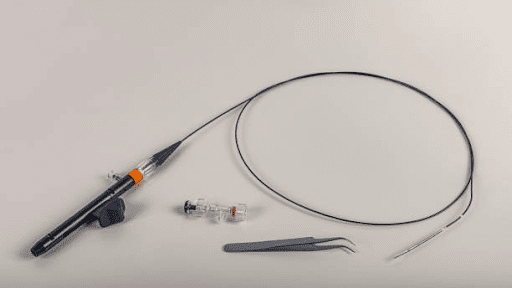
Accurate gastrointestinal diagnosis depends on reliable tools and standardized clinical workflows. Among these, the endoscope biopsy channel for sale plays a critical role in modern diagnostic procedures. At the same time, the H.pylori antigen test kit has become a widely accepted method for detecting Helicobacter pylori infections.
Medical buyers, hospitals, and diagnostic centers increasingly seek solutions that combine procedural accuracy with operational efficiency. Understanding how biopsy channels support diagnostic sampling and how antigen testing confirms infection is essential for informed purchasing decisions.
This article explores the relationship between endoscope biopsy channels and H.pylori antigen test kits from a buyer perspective. Without any further delay, let’s begin with our article!
What is an Endoscope Biopsy Channel?
An endoscope biopsy channel is a dedicated internal passage within an endoscope that allows medical instruments to be inserted and operated during an endoscopic procedure. This channel runs from the control section of the endoscope to its distal tip and is specifically designed to pass biopsy forceps, brushes, needles, or other therapeutic tools.
Through the biopsy channel, clinicians can collect tissue samples, remove small lesions, deliver fluids, or perform minor treatments while maintaining clear visualization. The channel is engineered with precise dimensions and smooth inner surfaces to ensure safe instrument movement, minimize friction, and reduce the risk of contamination. In diagnostic procedures, the endoscope biopsy channel plays a critical role by enabling accurate tissue sampling without the need for invasive surgery, supporting early disease detection and effective clinical decision-making.
Why Is an Endoscope Biopsy Channel for Sale Essential for Gastrointestinal Diagnosis?
An endoscope biopsy channel for sale enables precise tissue sampling during endoscopic procedures. It allows clinicians to insert biopsy forceps or sampling tools accurately. This functionality is essential when evaluating gastric mucosa for abnormalities.
High quality biopsy channels ensure smooth instrument passage without resistance. Poor channel construction can damage tools or compromise sample quality. Internal surface smoothness directly affects procedural reliability.
Material selection also influences performance. Medical grade polymers or stainless steel linings resist corrosion and repeated sterilization. This durability supports long term clinical use. Compatibility with standard endoscope models is another buying factor. Universal fitting reduces equipment redundancy and training requirements. Buyers should assess internal diameter consistency and channel length accuracy.
A dependable endoscope biopsy channel for sale improves diagnostic confidence. It supports consistent sampling outcomes and reduces procedural risks for patients and clinicians alike.
How Does the H.pylori Antigen Test Kit Support Accurate Infection Detection?
The H.pylori antigen test kit detects Helicobacter pylori antigens in stool samples. This method provides non-invasive diagnostic confirmation. It is widely used for initial screening and post treatment monitoring.
Test accuracy depends on antibody specificity and reagent stability. High quality kits deliver consistent sensitivity and specificity rates. This reliability reduces false negatives and unnecessary follow up procedures. Ease of use is another advantage. Most H.pylori antigen test kits require minimal laboratory equipment. Clear result interpretation supports faster clinical decisions.
Storage conditions and shelf life influence purchasing decisions. Kits with stable reagents reduce waste and inventory pressure. Buyers should review validation studies and regulatory approvals.
When paired with endoscopic evaluation, antigen testing strengthens diagnostic certainty. It offers confirmation without additional invasive sampling. This balance improves patient comfort and diagnostic efficiency.
How Are Endoscope Biopsy Channels Used Alongside H.pylori Antigen Test Kits?
Endoscopic biopsy and antigen testing serve complementary diagnostic roles. During endoscopy, the biopsy channel allows direct visualization and targeted sampling. This step identifies mucosal changes associated with infection.
However, biopsy results alone may not provide complete infection confirmation. The H.pylori antigen test kit verifies active infection status. This confirmation is valuable when histological findings are inconclusive.
Using both methods improves diagnostic accuracy. Endoscopy evaluates structural abnormalities. Antigen testing confirms bacterial presence. This dual approach supports evidence based treatment decisions.
From a procurement perspective, compatibility matters. Biopsy channels must support efficient sampling without prolonging procedures. Antigen kits should integrate easily into laboratory workflows.
Healthcare providers benefit from coordinated diagnostic tools. Combining reliable biopsy channels with validated antigen kits enhances workflow efficiency and patient outcomes.
What Quality Factors Should Buyers Consider When Purchasing Diagnostic Components?
Quality evaluation begins with material safety and certification. An endoscope biopsy channel for sale must meet medical device standards. Compliance ensures patient safety and regulatory acceptance.
Manufacturing precision affects performance. Uniform internal surfaces reduce friction and contamination risk. Buyers should request technical specifications and quality assurance documentation. Sterilization compatibility is essential. Channels must withstand repeated disinfection cycles without degradation. This feature reduces replacement frequency and operating costs.
For the H.pylori antigen test kit, batch consistency is critical. Reliable results depend on stable reagent formulation. Buyers should verify lot traceability and quality control protocols.
Supplier transparency strengthens purchasing confidence. Clear product documentation and responsive support reduce operational disruptions. Quality focused procurement protects diagnostic accuracy and institutional reputation.
How Can Medical Buyers Source Endoscope Biopsy Channels and Antigen Test Kits Effectively?
Effective sourcing starts with supplier evaluation. Buyers should assess manufacturing experience and product specialization. Dedicated diagnostic manufacturers often provide higher consistency.
Volume requirements influence pricing and availability. Bulk purchasing agreements may reduce costs for high demand facilities. However, quality should remain the primary consideration. Logistics and lead times also matter. Delayed supply can disrupt diagnostic services. Reliable distribution networks ensure uninterrupted clinical operations.
Technical support is another key factor. Suppliers should offer guidance on installation, usage, and storage. This support reduces training gaps and operational errors. Medical buyers benefit from aligning procurement strategies with clinical needs. Selecting a dependable endoscope biopsy channel for sale and a validated H.pylori antigen test kit ensures diagnostic reliability and long term value.
Conclusion:
Investing in accurate diagnostic tools is essential for effective gastrointestinal healthcare delivery. An endoscope biopsy channel for sale supports precise tissue sampling and procedural reliability. At the same time, the H.pylori antigen test kit provides non-invasive confirmation of active infection.
Together, these tools strengthen diagnostic accuracy and clinical confidence. Buyers who understand material quality, manufacturing precision, and supplier reliability make better purchasing decisions.
Coordinated sourcing reduces operational risks and supports consistent patient outcomes. By prioritizing certified products and dependable suppliers, healthcare facilities can optimize diagnostic workflows. This approach ensures that diagnostic accuracy, patient safety, and cost efficiency remain aligned with modern clinical standards.